🌿 Distillation and Solvent Extraction in Perfumery: How Scents Are Crafted?
✨ Introduction: Where Scent Begins
Behind every fine perfume lies a carefully crafted extraction process that transforms raw natural materials into fragrant ingredients. While many people focus on the finished bottle, few realize that the soul of a fragrance is born much earlier—during distillation or solvent extraction. These two techniques are the backbone of modern perfumery, shaping how flowers, woods, resins, and spices release their aromatic essence.
In this article, we’ll explore how distillation and solvent extraction work, why perfumers choose one method over the other, and how each technique influences scent quality, longevity, and character.
🔥 Distillation: The Art of Purifying Aroma
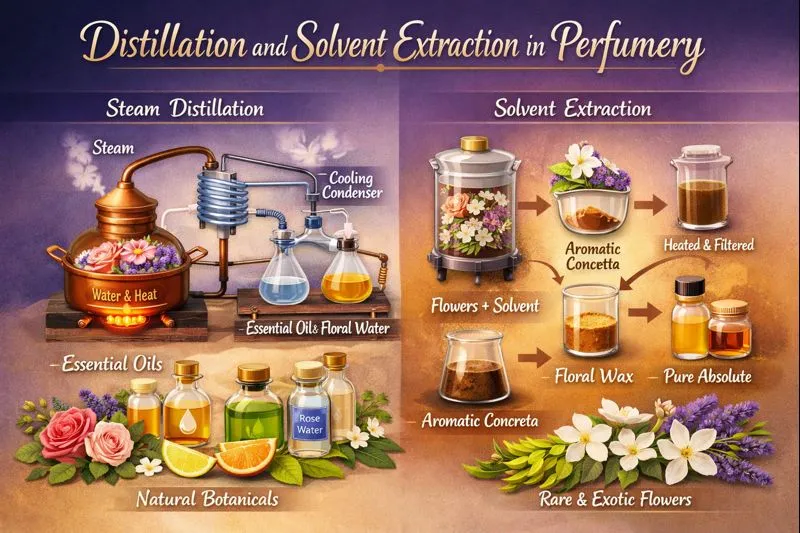
Distillation is one of the oldest fragrance extraction methods, used for centuries to capture essential oils from plants. It relies on heat and water to gently release aromatic molecules.
🌊 How Distillation Works
Plant material—such as lavender, rose petals, or sandalwood—is exposed to steam or boiling water. Heat causes aromatic compounds to evaporate along with the steam. Once cooled, the vapor condenses back into liquid, separating into two layers:
- Essential oil (the concentrated aromatic component)
- Hydrosol (aromatic water, such as rose water)
This process is widely used because it is relatively simple, natural, and preserves the purity of many ingredients.
🌿 Common Types of Distillation
- Steam distillation – Most common method, ideal for herbs, woods, and spices
- Water distillation – Traditional method often used for delicate flowers
- Vacuum distillation – Uses lower temperatures to protect fragile aromas
✅ Why Perfumers Love Distillation
- Produces clean, sharp, and transparent scents
- Ideal for essential oils used in both niche and mainstream perfumes
- Free from chemical solvents
However, not all botanicals survive heat well. Some flowers lose complexity or break down during distillation—this is where solvent extraction shines.
🧪 Solvent Extraction: Capturing Delicate Beauty

Solvent extraction was developed to handle fragile materials that cannot withstand high temperatures. This method excels at preserving the true scent of flowers like jasmine, tuberose, and violet.
🌸 How Solvent Extraction Works
Plant material is washed with a solvent (traditionally hexane or similar compounds). The solvent dissolves aromatic molecules, waxes, and pigments. Once the solvent evaporates, the result is a waxy substance called concrete.
The concrete is then treated with alcohol, separating pure aromatic compounds known as absolute.
🌺 Why Absolutes Matter in Perfumery
Absolutes are prized for their richness and depth. They often smell closer to the living flower than distilled oils, making them essential in luxury and artistic fragrances.
⚠️ Considerations of Solvent Extraction
- More expensive and time-consuming
- Requires careful solvent removal
- Produces highly concentrated, potent materials
Despite these challenges, solvent extraction remains irreplaceable for certain iconic perfume ingredients.
⚖️ Distillation vs. Solvent Extraction: A Clear Comparison
| Feature | Distillation | Solvent Extraction |
|---|---|---|
| Heat Used | Yes | No |
| Best For | Herbs, woods, spices | Delicate flowers, resins |
| Final Product | Essential oil | Absolute |
| Scent Profile | Clean, airy | Rich, deep, true-to-life |
| Cost | Moderate | Higher |
| Longevity in Perfume | Moderate | Often stronger |
This comparison explains why many perfumes combine ingredients extracted using both methods to achieve balance and complexity.
🧠 How Extraction Methods Shape the Final Scent
The extraction technique doesn’t just capture aroma—it defines personality.
- Distilled oils often feel fresh, transparent, and uplifting, making them ideal for top notes.
- Absolutes from solvent extraction feel sensual, dense, and emotional, perfect for heart and base notes.
A rose distilled as essential oil may smell light and green, while a rose absolute feels darker, warmer, and more romantic.
🌍 Modern Perfumery: Blending Tradition with Innovation
Today’s perfumers rarely rely on a single extraction method. Instead, they layer distilled oils with absolutes and modern aroma molecules to create multidimensional scents. Sustainability also plays a growing role, encouraging improved distillation efficiency and safer solvent alternatives.
Natural perfume lovers often seek transparency about extraction methods, while niche brands highlight traditional distillation or rare absolutes as marks of craftsmanship.
If you enjoy learning how raw materials are transformed into fragrance, you may also find it fascinating to explore “What Is Peru Balsam – And Why Is It Used in High-End Perfumes?”. That article dives deeper into one specific natural ingredient and shows how extraction methods influence its warm, resinous character in luxury scents.
🌟 Final Thoughts: Crafting Emotion from Nature
Distillation and solvent extraction are more than technical processes—they are creative tools that turn nature into emotion. Understanding these methods reveals why perfumes smell the way they do and why certain notes feel airy while others feel intoxicating.
Whether you prefer fresh essential oils or rich floral absolutes, every fragrance tells a story shaped by how its ingredients were extracted.
Do you personally prefer perfumes that feel light and transparent—or rich and deeply floral? Share your thoughts in the comments below 👇
Discover more from Perfume Cultures
Subscribe to get the latest posts sent to your email.